Enhanced oil recovery (EOR) by carbon dioxide (CO2) injection is an effective method for recovering additional oil beyond waterflooding. However, the CO2-EOR process is handicapped, especially in thick reservoirs, by CO2 gravity override. Because of density differences between the injected CO2 and resident fluids in the reservoir, the lighter CO2 tends to rise to the top of the reservoir, thereby bypassing some of the remaining oil. This paper investigates the use of gelling CO2/water emulsions, stabilized by silica nanoparticles, to control the mobility of CO2.
Introduction
The CO2-EOR process, while increasingly important, is handicapped by several challenges. The first challenge is the gravity override of the injected CO2 because of density differences between the injected CO2 and resident fluids in the reservoir. The second is viscous fingering caused by the lower viscosity of the injected CO2. Typical dense CO2 viscosity at reservoir conditions is in the range of 0.05–0.1 cp, which is much lower than the viscosity of resident oil and brine. The resulting unfavorable mobility ratio leads to viscous fingering, causing early CO2 breakthrough, high CO2-usage factors, poor sweep efficiency, and low overall oil recovery. The third challenge associated with CO2 EOR is reservoir geology and heterogeneities, including high-permeability streaks and fractures that can impact the sweep efficiency of a CO2-EOR flood. Most of these challenges have been countered by use of water-alternating-gas (WAG) processes. Although the WAG process improves the mobility of CO2, it loses its effectiveness in controlling CO2 mobility deep into the reservoir and beyond a certain distance from the wellbore.
This paper discusses another technique that provides an excellent alternative to WAG to control CO2 mobility: the CO2/foam emulsion. The main purpose of using CO2 foams is to increase the viscosity of the CO2 and therefore reduce its mobility through the reservoir. This emulsion is generated with water-soluble surfactants, CO2-soluble surfactants, or nanoparticles. This paper investigates the use of gelling CO2/water emulsions, stabilized by silica nanoparticles, to control the mobility of CO2.
Experimental Studies
Materials. Fumed and colloidal silica nanoparticles were used in this study. These nanoparticles have a silylating agent that renders them CO2-philic.
The chemical-screening experiments were conducted to explore the possibility of generating both water-in-CO2 (W/C) and CO2-in-water (C/W) emulsions. Iso-octane (I-C8) was used as a proxy fluid to represent supercritical CO2 (SC CO2) in the screening experiments. I-C8 has density and solvency characteristics comparable with those of SC CO2. Many noted experts have used I-C8 to represent SC CO2 in the area of CO2 EOR. The water used to prepare different emulsion systems was seawater with a salinity of 57,000 ppm. CO2 was locally supplied and had 99.99% purity. It was compressed to 5,000 psi in high-pressure titanium cylinders and used as SC CO2.
Foam-Stability Test. Two types of emulsions were prepared on the basis of the degree of nanoparticle hydrophobicity. The first type was prepared by emulsifying water in I-C8 containing hydrophobic particles at different weight percentages, while the other type was prepared by emulsifying I-C8 in water containing hydrophilic nanoparticles. The nanoparticle weight percentage varied between 0.5 and 2 wt%, and the I-C8-phase volume varied between 50 and 90%. The morphology of stable emulsion (i.e., the dispersed-phase size and size distribution) was characterized by using an optical microscope fitted with a digital camera. This test was performed in two stages: room and reservoir conditions.
The high-pressure experiments were conducted with CO2 and involved the use of specialized equipment able to handle pressure and temperature as well as having a viewing window for the observation of foam.
Viscosity Measurement
Following the stability screening tests, several experiments were conducted to investigate the increase of CO2 viscosity after generating water/CO2 emulsions using different nanoparticles. These experiments were conducted with a flow loop (Fig. 1). This flowloop is equipped with two accumulators, two pumps with a maximum pressure of 5,000 psi, a horizontal pipe section with an inside diameter of 6.99×10–4 m and a length of 1.5 m, a mixing loop, two differential-pressure transducers, and an oven. A backpressure of approximately 1,800 psi and a temperature of 200°F were maintained in all experiments.
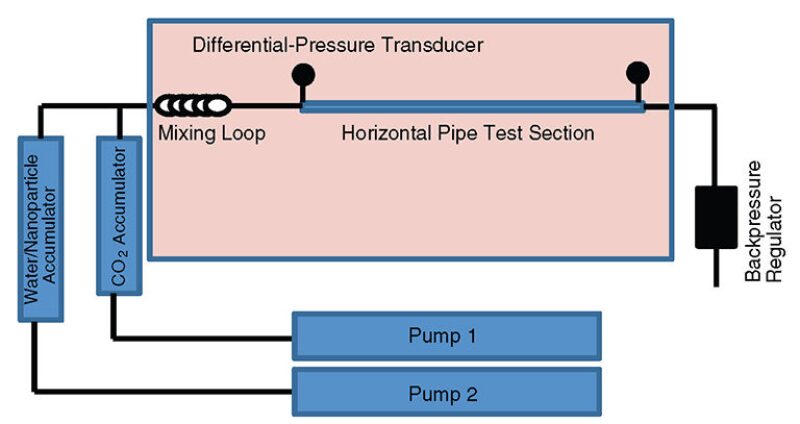
Different water/CO2 emulsions were injected into the flow-loop apparatus, and pressure values across the horizontal pipe section were recorded. These emulsions were generated by use of different water/CO2 volume ratios and with nanoparticle concentrations of 0.5, 1, and 2 wt%. To generate a certain water/CO2 emulsion system, liquid CO2 and nanoparticle solutions of desired concentrations were pumped by use of two separate pumps. These two fluids were mixed in the mixing loop, where the emulsion was generated. With a total injection rate of 20 cm3/min, the volume percentage of both CO2 and water in the emulsion system was varied by controlling the injection rate of both water and CO2.
Results and Discussions
Several experiments were conducted to explore the possibility of generating both W/C and C/W emulsions with either colloidal or fumed silica nanoparticles. The volume ratios between I-C8 and water were varied. The I-C8/water emulsion-generation experiments were performed by use of two main silica-nanoparticle systems: fumed silica and hydrophilic colloidal silica.
Foam Generation. Water/I-C8 emulsions were generated using two types of fumed silica nanoparticles. The nanoparticles are 100% hydrophobic, with equivalent surface areas of 200 m2/g. One type has a particle size of 16 nm, while the other has a 12-nm particle size. The concentration of these two silica nanoparticles was varied between 0.5 and 2 wt%. In addition, the I-C8 volume percentage ranged between 50 and 90%. Initially, water/I-C8 emulsions were generated successfully at different nanoparticle concentrations of 0.5 to 2 wt% and I-C8 volume percentages of 50, 70, and 90%. The average size of water droplets in these emulsions generated by use of either type of nanoparticle was found to be nearly 35 µm. These results indicated that nanoparticle size had little or no effect on water-droplet size in the water/I-C8 emulsions. The stability results for water/I-C8 emulsions generated using either type of nanoparticle are comparable. Given the fact that the two types of nanoparticle have different particle sizes, it can be concluded that emulsion stability was not affected by nanoparticle size.
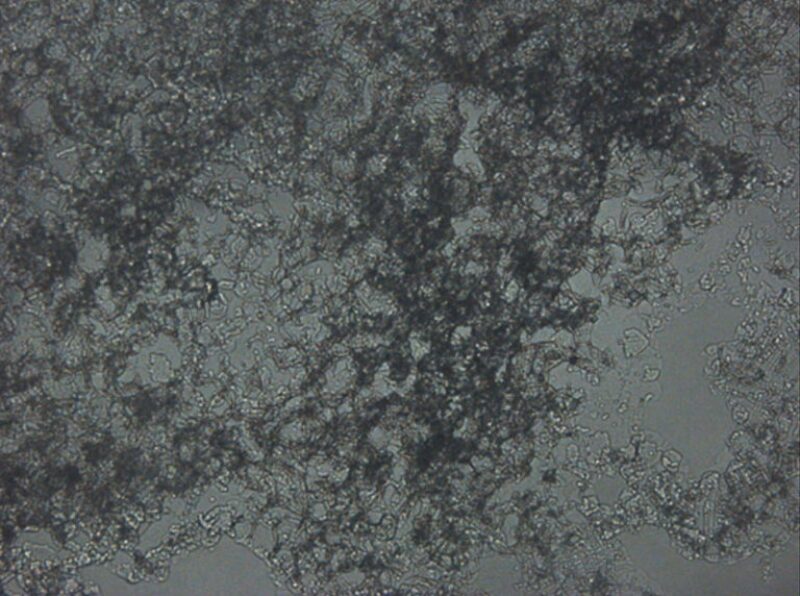
The stable-emulsion height percentage of the water/I-C8 emulsion increased as the volume ratio decreased in the emulsion system. The nanoparticle concentration also affected the stability of water/I-C8 emulsions. Increasing the nanoparticle concentration resulted in a more-stable emulsion system for the 90:10 I-C8/water emulsion. Another interesting phenomenon observed in these emulsions is the formation of gel because of the interaction of silica nanoparticles, as shown in Fig. 2. The formation of gels should assist further in controlling CO2 mobility during CO2-EOR applications.
Effect of Hydrophobicity Degree and Silica Type. To assess the effect of both hydrophobicity degree and silica type on the water/I-C8 emulsions, another set of experiments was conducted using a colloidal silica. These silica nanoparticles are 100% hydrophilic, with an average particle size of 22 nm and a surface area of 140 m2/g.
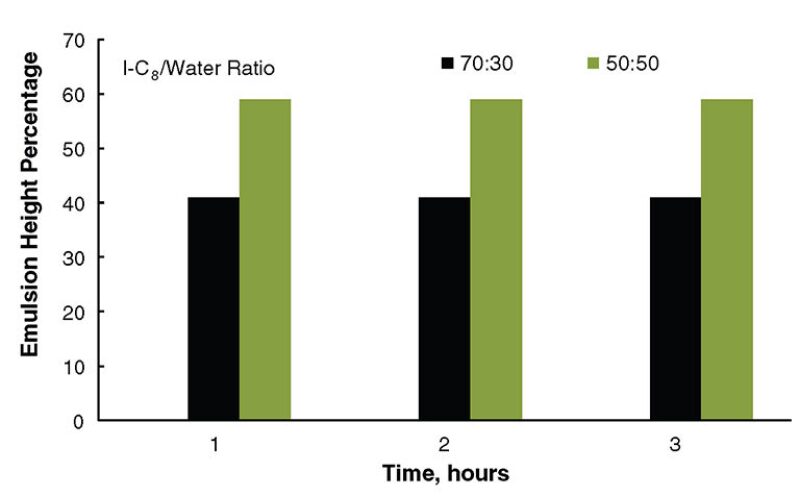
I-C8/water emulsions were generated with the colloidal silica nanoparticles. The emulsions formed using these nanoparticles were not stable. At 90% I-C8 volume percentage, no emulsions were generated even when the colloidal-silica-nanoparticle concentration was increased to 2 wt%. Relatively speaking, the I-C8/water emulsions were more stable at lower I-C8 volume percentage (Fig 3). For example, the stable-emulsion height percentage was 35 and increased to 58 when the I-C8 volume percentage was decreased from 70 to 50% after 2 hours. The phenomenon of gelled emulsions was also noted in the I-C8/water emulsions. The degree of gelation was stronger at higher nanoparticle concentrations.
Foam Viscosity. The emulsion viscosity was measured using the flow loop described previously. The viscosity, at a fixed flow rate, is approximated with the pressure drop across the flow loop. In reality, emulsions are known to be non-Newtonian fluids; their viscosity is dependent on shear rate. The CO2/water emulsions are also compressible, and their density is dependent on applied pressure.
At constant flow rate, many empirical equations have been proposed to correlate fluid-flow behavior to its physicochemical properties, such as density and viscosity. For a discussion of some of these equations, please see the complete paper.
The maximum theoretical emulsion density was 634.8 kg/m3 and the maximum obtained Reynolds number was 147.9, confirming that all experiments were conducted under laminar flow. CO2/water emulsion viscosity using both fumed silica nanoparticles was comparable and had similar profiles, indicating that nanoparticle size had no significant effect on the rheological properties of the generated emulsion.
At constant CO2/water volume ratio, the emulsion viscosity increased when nanoparticle concentration was increased. The highest increase in the viscosity of CO2/water emulsion attributable to the increase of nanoparticle concentration was noticed at a 50:50 CO2/water volume ratio. When the nanoparticle concentration was increased from 0.5 to 2 wt%, the CO2/water emulsion viscosity increased by 40, 55, and 83% at 90:10, 70:30, and 50:50 CO2/water volume ratios, respectively.
At constant nanoparticle concentrations, it was noticed that the highest emulsion viscosity occurred at a 50:50 CO2/water volume ratio. The highest effect of increasing the CO2/water volume ratio was noticed at a nanoparticle concentration of 2 wt%. An increase of 60% in the CO2/water emulsion viscosity was noticed at 2 wt% of nanoparticles when the CO2/water volume ratio was decreased from 90:10 to 50:50.
Conclusions
Several experiments were conducted to explore the possibility of generating both W/C and C/W emulsions using either colloidal or fumed silica nanoparticles. In these experiments, the immiscible two-phase water/I-C8 fluid was used to represent a water/CO2 system. The main conclusions from these experiments include the following:
- Stable water/I-C8 emulsions were generated using different concentrations of hydrophobic silica nanoparticles and different water/I-C8 volume percentages.
- The nanoparticle concentrations and water/I-C8 percentages had an impact on the stability of formed emulsions of I-C8 and water.
Another set of experiments was conducted using liquid CO2 to explore the possibility of generating CO2/water emulsions and to investigate the rheological properties of these emulsions. The main conclusions of these experiments include the following:
- Stable C/W and W/C emulsions were generated at different CO2/water volume ratios using different concentrations of hydrophilic and hydrophobic silica nanoparticles, respectively.
- Generating CO2/water emulsion was very effective in enhancing the viscosity of liquid CO2.
- At constant CO2/water volume ratio, the emulsion viscosity increased when the nanoparticle concentration was increased.
- At constant nanoparticle concentration, the emulsion viscosity increased when the CO2 volume percentage was decreased.
This article, written by JPT Technology Editor Chris Carpenter, contains highlights of paper SPE 166072, “Gelled Emulsions of CO2/Water/Nanoparticles,” by Fawaz M. Al-Otaibi, Sunil L. Kokal, Yun C. Chang, Jassi F. Al-Qahtani, and Amin M. Al-Abdulwahab, Saudi Aramco, prepared for the 2013 SPE Annual Technical Conference and Exhibition, New Orleans, 30 September–2 October. The paper has not been peer reviewed.
