Ferrous sulfide (FeS) precipitation increases during shale-gas and -oil production because of increased biologically and thermally induced sulfide production. Although FeS scale is abundant, little is understood about its precipitation and inhibition properties. A plug-flow apparatus adds reliable data to the limited database of scaling kinetics in flowing pipes, and it provides a new method to study the effect of inhibitors in oil-production systems.
Introduction
During oil production, mineral-salt precipitation (scale) builds up inside wellbores. It is very difficult to study the formation and inhibition of FeS because the experiments must be performed under rigorous anoxic conditions and the induction time is very short. Compared with common oilfield scales, FeS scale has distinctive characteristics. FeS can cause galvanic corrosion in the presence of water. FeS has several crystalline forms with different sulfur/iron ratios that result in different solubilities of FeS. Additionally, FeS particles are oil-wet and can be coated easily with oil or hydrocarbons; these coatings can act as a diffusion barrier that retards acid reaction with the scale.
To evaluate the risk of scaling in pipes, a plug-flow apparatus was used to investigate CaCO3-scale precipitation on stainless-steel tubes. In the study of CaCO3, a precoated plug-flow reactor provided a constant composition at the tubing exit, including constant pH, saturation index (SI), ionic strength, and surface area. The plug-flow system provided a feasible approach to study the kinetics of initial attachment and crystal growth. FeS had not been studied with a plug-flow system; therefore, it was important to develop a plug-flow setup to investigate FeS formation and precipitation, enhancing the limited database of scaling kinetics in flowing pipes.
To eliminate the influence of scale on oil production, scale inhibitors often are needed to minimize the scale buildup. The controlling method of scale inhibitors is to delay, reduce, or prevent scale formation. Scale inhibitors can prevent further growth of scale crystals by adsorbing onto the surface of the crystals. In addition to scale inhibitors, dispersants and chelating agents are applied to sequester metal cations, preventing them from reacting with anions.
The objectives of this research were to investigate the effects related to the driving force on FeS precipitation in batch and plug-flow systems, and to conduct a series of laboratory studies on scale-inhibition approaches for FeS by use of commercial scale inhibitors and chelating agents.
Materials and Method
All experiments were conducted under anoxic conditions in the presence of a reducing agent (hydroxylamine hydrochloride) and sparged by 99.9% argon gas. To purge the reaction vessel of the batch reactor system, argon gas was passed through an oxygen-scavenger trap and saturated with deionized water; the concentration of dissolved oxygen was less than 5 ppb. A second-order kinetics model (detailed in the complete paper) was used to interpret the nucleation and precipitation results for the batch reactor.
In the plug-flow system shown in Fig. 1, a precoated plug-flow reactor was developed to study FeS precipitation kinetics on a carbon-steel tube over the temperature range of 20–50°C. The characteristics of the carbon-steel tube are listed in Table 1 of the complete paper.
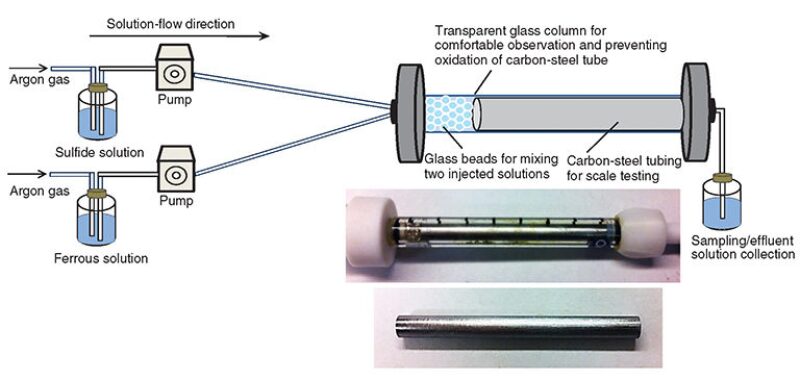
Inhibiting FeS Precipitation. For inhibition experiments, two scale inhibitors, polyphosphono carboxylic acid (PPCA) and polyvinyl sulfonate (PVS), and five dispersants/chelating agents—sodium citrate; ethylenediamine-tetraacetic-acid (EDTA) disodium salt dihydrate; deferoxamine mesylate salt; pentetic acid; and 1,10-phenanthroline—were used in the batch system. Different concentrations of these inhibitors were studied.
Results
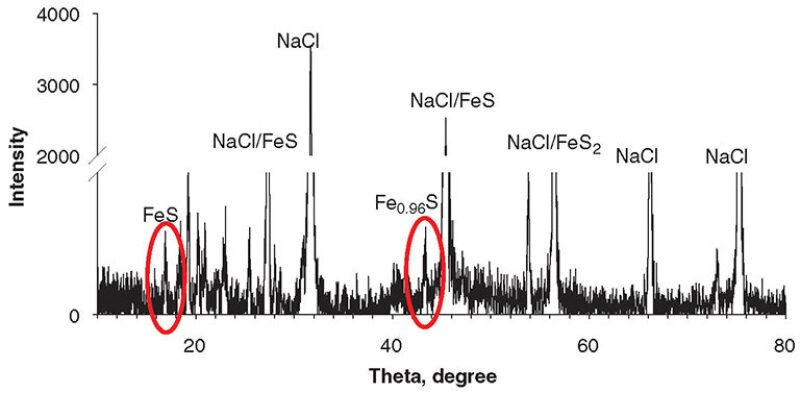
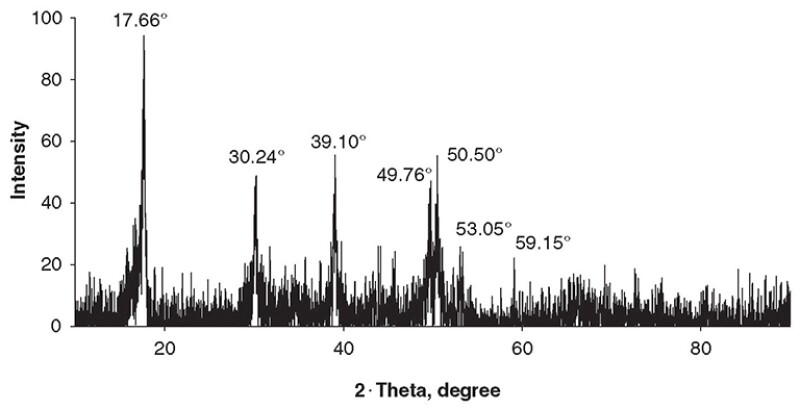
FeS Characterization. The physicochemical examinations of powders of the solid precipitates by X-ray diffraction (XRD), Brunauer-Emmett-Teller technique, scanning electron microscope (SEM), and X-ray photoelectron spectroscopy showed that FeS formed under all supersaturation conditions. Fig. 2 shows the XRD patterns of products, which were stoichiometrically consistent with FeS, Fe0.96S, and FeS2, for the batch system. These observed FeS phases were consistent with peak positions; however, the relative low peak intensities of FeS compared with the high-intensity peaks of sodium chloride resulted from the pre-existing sodium chloride in the prepared brine solution in the batch system. As shown in Fig. 3, for the plug-flow system, XRD analysis revealed the structure and composition of the FeS particles, with peaks of FeS at 17.66, 30.24, 39.10, 49.76, 50.5, 53.05, and 59.15°, as expected for mackinawite. Fig. 4 above shows a typical SEM image of the FeS aggregate from the batch system. The morphology revealed the amorphous structure of synthesized FeS particles in the batch reactor. Fig. 5 shows an SEM image of the FeS aggregate formed on the inner surface of the carbon-steel tube compared with the smooth inner surface of the carbon-steel tube.

Kinetics of FeS Precipitation in the Batch System. Supersaturation Effect. Experiments were performed over a range of supersaturations and are summarized in Table 2 in the complete paper. Immediately after adding sulfide solution into the reactor containing ferrous ions, a very fast precipitation occurred, consistent with previous studies that showed that the rate of FeS precipitation is essentially instantaneous. As the SI value increased from 0.62 to 1.43 by increasing the concentration of FeCl2 and Na2S, the rate constant increased from 700.7 to 10 874.3 L/mol⋅min. The higher SI also resulted in higher fractional usage of reactants, leading to greater production of FeS. The increased supersaturation is the result of increasing the initial concentration of reactants.
Temperature Effect. The rate of FeS precipitation was investigated at temperatures of 4, 25, 50, 65, and 75°C at a constant pH of 6.75±0.05. The second-order kinetics model was sufficient to represent the experimental data at 4, 25, and 50°C. At 50°C and below, nucleation of FeS reached equilibrium in approximately 30 minutes, and the rate constant increased from 158.2 L/mol⋅min at 4°C to 1974.9 L/mol⋅min at 50°C, and the SI increased from 0.57 at 4°C to 0.61 at 50°C. However, for higher temperatures, 65 and 75°C, the nucleation results did not follow the second-order model, and the SI decreased from 0.61 at 50°C to 0.60 at 75°C.
Effect of pH. The pH value of the reaction solution was adjusted by adding NaOH or HCl. The results of the effect of pH on the precipitation of FeS are shown in Fig. 6c and Table 2 in the complete paper. The temperature was controlled at 50°C, and the pH was adjusted at 6.40, 6.75, and 7.10, which means the amount of hydroxide ion OH− increased by approximately five times. The kinetics constant increased from 1068.2 to 5000 L/mol⋅min as the pH increased from 6.40 to 7.10, showing that the rate is directly proportional to the OH− concentration.
Kinetics of FeS Precipitation in a Plug-Flow System. Temperature Effect. The rate of FeS precipitation was investigated at temperatures of 22.6, 35, and 50°C at a constant pH of 6.05±0.05. A pseudofirst-order model was found to be sufficient to represent the experimental data of 4, 22.6, and 50°C. An increase in temperature resulted in an increased rate of FeS precipitation in the plug-flow system and in the batch system. The mass-transfer coefficient increased from 0.0029 cm/s at 22.6°C to 0.0045 cm/s at 50°C (a 1.55‑fold increase), and the SI increased from 0.153 at 22.6°C to 0.225 at 50°C (a 1.47-fold increase).
Effects of Ionic Strength and pH. The effect of ionic strength on the precipitation of FeS was studied at 50°C, and results are shown in Fig. 7b and Table 3 in the complete paper. It was observed that the rate constants of FeS precipitation decreased when the electrolyte concentration was increased from 0.5- to 3-M NaCl solution. However, because the formation speed of FeS is nearly instantaneous, the inhibitory effect of NaCl with a high concentration of FeS was observable only at the beginning of nucleation (less than 30 seconds). Similar results were found in a previous study—cations had an inhibitory effect on mineral nucleation and precipitation.
The temperature was controlled at 50°C, and pH was adjusted to be 5.85, 6.05, and 6.45. The mass-transfer coefficient increased from 0 to 0.0066 cm/s as pH increased from 5.85 to 6.45. Increasing the pH increased the precipitation rate and ferrous-ion usage. The increase of pH yielded an increased SI, which increased the rate of FeS precipitation, as expected.
The pH had a strong effect on the particle-size distribution of the final precipitation product, and precipitation formed at higher pH and had a shorter induction time. High pH values and relatively high supersaturation values favored precipitation of smaller FeS particles.
Inhibition of FeS. Inhibiting FeS precipitation was investigated by use of two common scale inhibitors and five chelating agents in the batch reactor. All experiments were conducted in 2-M NaCl solutions at a constant SI, pH of 6.75, and 50°C. A slight inhibition of FeS nucleation was observed in the presence of PPCA with a concentration of 3600 mg/L, compared with the experiment without an inhibitor.
The PPCA adsorbed slowly onto the surface of FeS crystal to prevent FeS precipitation. PVS showed almost no inhibition of FeS precipitation. This lack of inhibition might result from no, or little, adsorption of PVS onto FeS.
Sodium citrate and other chelating agents were studied. Sodium citrate concentrations used in this study were from 73 to 2200 mg/L. Significant inhibitions of FeS nucleation were observed during the 30-minute reaction time at all sodium citrate concentrations. These results indicate that sodium citrate acts as a modest threshold inhibitor of FeS precipitation. In addition, 1,10-phenanthroline and EDTA, pentetic acid, and deferoxamine were used to investigate the inhibitory effects of these chelating agents. EDTA was a very effective stoichiometric chelating inhibitor of FeS. Compared with sodium citrate, EDTA at 73 mg/L was an effective inhibitor of FeS nucleation. Pentetic acid, deferoxamine, and 1,10-phenanthroline also showed effective inhibition on FeS nucleation at 59, 47, and 200 mg/L.
Conclusions
The nucleation and precipitation kinetics of FeS in NaCl solutions depend on solution supersaturation, temperature, ionic strength, and pH. A plug-flow system was developed that used an FeS-precoated carbon-steel tube, which provided a constant composition and experimental conditions. The FeS precipitation can be described as a pseudofirst-order diffusion-controlled process. The mass-transfer coefficient increased with temperature and pH, and decreased with ionic strength. Experiment results indicated that pH is the most influential factor on FeS formation. Applying a diffusion-kinetics model to the thermodynamic scaling predictions could help manage FeS-scaling problems. Commercial scale inhibitors (PPCA and PVS) did not exhibit significant inhibitory effects on FeS precipitation. Sodium citrate, EDTA, and other chelating agents were more effective at inhibiting FeS nucleation.
This article, written by Dennis Denney, contains highlights of paper SPE 169748, “Kinetics and Inhibition of Ferrous Sulfide Nucleation and Precipitation,” by Qiliang Wang, Zhang Zhang, Amy Kan, SPE, and Mason Tomson, SPE, Rice University, prepared for the 2014 SPE International Oilfield Scale Conference and Exhibition, Aberdeen, 14–15 May. The paper has not been peer reviewed.

